

Why does NH3 form hydrogen bond but PH3 does not?
Nitrogen has an electronegativity value 3.0, which is much higher than that of H (2.1). As a result, N – H bond is quite polar and hence NH3 undergoes intermolecular H – bonding.
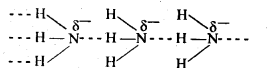
Phosphorus have an electronegativity value 2-1. Thus, P – H bond is not polar and hence PH3 does not undergo H – bonding.
Justify the placement of O, S, Se, Te and Po in the same group’of the periodic table in terms of electronic configuration, oxidation state and hydride formation.
Assertion (A): HNO3 makes from passive.
Reason (R): HNO3 forms a protective layer of ferric nitrate on the surface of iron.
Assertion (A): HI cannot be prepared by the reaction of KI with concentrated H2SO4.
Reason (R): HI has lowest H – X bond strength among halogen acids.
How is nitrogen prepared in the laboratory? Write the chemical equations of the reactions . involved.
In the ring test of NO3 ion.Fe2+ion reduces nitrate ion to nitric oxide, which combines with Fe2+ (aq.) ion to form brown complex. Write the reactions involved in the formation of brown ring.
PCl5 reacts with finely divided silver on heating and a white silver salt is obtained, which dissolves on adding excess aqueous NH3 solution. Write the reactions involved to explain what happens.
Write balanced equations for the following:
(i) NaCl is heated witlrsulphuric acid in the presence of MnO2
(ii) Chlorine gas is passed into a solution of Nal in water.
Give the formula and describe the structure of a noble gas species which is isostructural with: (i) ICI4– (ii) IBr2– (iii) Br03–
Comment on the nature of two S-O bonds formed in S02 molecule. Are the two S-O bonds in this molecule equal ?
Knowing the electron gain enthalpy values of O—>O– and O—>O2- as -141 and 702 kJ mol-1 respectively, how can you account for [he formation of a large number of oxides having O2- species and not O–?
Write balanced equations for the following:
(i) NaCl is heated witlrsulphuric acid in the presence of MnO2
(ii) Chlorine gas is passed into a solution of Nal in water.
In a cyclotrimeta phosphoric acid molecule, how many single and double bonds are present? .
(a) 3 double bonds; 9 single bonds
(b) 6 double bonds; 6 single bonds
(c) 3 double bonds; 12 single bonds
(d) Zero double bonds; 12 single bonds
On heating with concentrated NaOH solution in an inert atmosphere of CO2, white phosphorus gives a gas. Which of the following statement is incorrect about the gas?
(a) It is highly poisonous and has smell like rotten fish.
(b) Its solution in water decomposes in the presence of light.
(c) It is more basic than NH3
(d) It is less basic than NH3
Which of the following acid forms three series of salts?
(a) H2PO2 (b) H3BO3 (C)H3PO4(d)H3PO3
Which of the following options are not in accordance with the properly mentioned against them?

Which of the following statements are correct?
(a) Among halogens, radius ratio between iodine and fluorine is maximum.
(b) Leaving F – F bond, all halogens have weaker X – X bond than X – X’ bond in interhalogens.
(c) Among interhalogen compounds maximum number of atoms ate present in iodine fluoride.
(d) Interhalogen compounds are more reactive than halogen compounds.
Which of the following statements are correct?
(a) All three N – O bond lengths in HNO3 are equal.
(b) All P – Cl bond lengths in PCl5 molecule in gaseous state are equal.
(c) P4 molecule in white phosphorus have angular strain therefore white phosphorus is very reactive.
(d) PCl5 is ionic in solid state in which cation is tetrahedral and anion is octahedral.
Which of the following statements are correct?
(a) S – S bond is present in H2S2O6.
(b) In peroxosulphuric acid (H2SO5) sulphur is in +6 oxidation state.
(c) Iron powder along with Al2O3 and K2O is used as a catalyst in the preparation of NH3 by Haber's process
(d) Change in enthalpy is positive for the preparation of SO3 by catalytic oxidation of SO2.
Why is nitric oxide paramagnetic in gaseous state but the solid obtained on cooling is diamagnetic?
P4O6 reacts with water according to equation P4O6 + 6H2O Calculate the volume of 0.1 M NaOH solution required to neutralize the acid formed by dissolving 1.1 g of P4O6 in H2O.
Nitric acid forms an oxide of nitrogen on reaction with P4O10. Write the reaction involved. Also write the resonating structures of the oxide of nitrogen formed.
Phosphorus forms a number of oxoacids. Out of these oxoacids phosphinic acid has strong reducing property. Write its structure and also write a reaction showing its reducing behaviour.
Match the compounds given in Column I with the hybridization and shape given in Column II and mark the correct option.
