

Why is bond angle in PH+4 ion higher than in PH3 ? (Pb. Board 2009)
In both PH3 and PH+4 ion, the phosphorus atom is sp3 hybridised. However, in PH3 the central atom has apyramidal structure due to the presence of lone electron pair on the phosphorus atom.
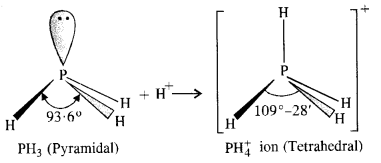
Because of lone pair : shared pair repulsion which is more than that of shared pair : shared pair repulsion, the bond angle in PH3 is nearly 93-6°. In PH+4 ion, there is no lone electron pair on the phosphorus atom. It has a tetrahedral structure with bond angle of 109°-28′. Thus, the bond angle in PH+4 ion is higher than in PH3.
Assertion (A): HNO3 makes from passive.
Reason (R): HNO3 forms a protective layer of ferric nitrate on the surface of iron.
Justify the placement of O, S, Se, Te and Po in the same group’of the periodic table in terms of electronic configuration, oxidation state and hydride formation.
In the ring test of NO3 ion.Fe2+ion reduces nitrate ion to nitric oxide, which combines with Fe2+ (aq.) ion to form brown complex. Write the reactions involved in the formation of brown ring.
Assertion (A): HI cannot be prepared by the reaction of KI with concentrated H2SO4.
Reason (R): HI has lowest H – X bond strength among halogen acids.
How is nitrogen prepared in the laboratory? Write the chemical equations of the reactions . involved.
Give the formula and describe the structure of a noble gas species which is isostructural with: (i) ICI4– (ii) IBr2– (iii) Br03–
Nitric acid forms an oxide of nitrogen on reaction with P4O10. Write the reaction involved. Also write the resonating structures of the oxide of nitrogen formed.
PCl5 reacts with finely divided silver on heating and a white silver salt is obtained, which dissolves on adding excess aqueous NH3 solution. Write the reactions involved to explain what happens.
Why does nitrogen show catenation properties less than phosphorus ? (C.B.S.E. Foreign 2009)
Write balanced equations for the following:
(i) NaCl is heated witlrsulphuric acid in the presence of MnO2
(ii) Chlorine gas is passed into a solution of Nal in water.
With which neutral molecule is ClO– isoelectronic? Is this molecule Lewis acid or base ? (Pb. Board 2009)
Comment on the nature of two S-O bonds formed in S02 molecule. Are the two S-O bonds in this molecule equal ?
The HNH angle value is higher than HPH, H AsH and HSbH angles. Why?
(Hint: Can be explained on the basis of sp3 hybridisation in NH3 and only s-p bonding , between hydrogen and other elements of the group).
Knowing the electron gain enthalpy values of O—>O– and O—>O2- as -141 and 702 kJ mol-1 respectively, how can you account for [he formation of a large number of oxides having O2- species and not O–?