

A flask was heated from 27 °C to 227 °C at constant pressure. Calculate the volume of the flask if 0.1 dm3 of air measured at 227 °C was expelled from the flask.
Let the volume of the flask = V dm3 (after expelling the air)
V1 = V dm3, T1 = 27 + 273 = 300K
VT2 = (V + 0.1) dm3, T2 = 227 + 273 = 500 K
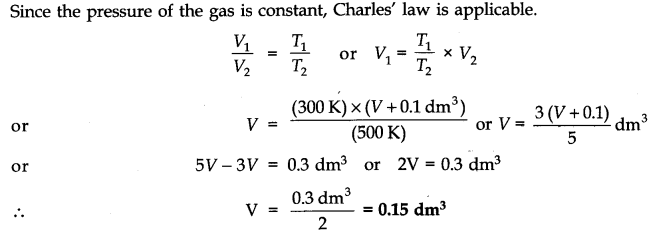
Assertion (A): Gases do not liquefy above their critical temperature, even on applying high pressure.
Reason (R): Above critical temperature, the molecular speed is high and intermolecular attractions cannot hold the molecules together because they escape because of high speed.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
Which of the following changes decrease the vapour pressure of water kept in a sealed vessel?
(a) Decreasing the quantity of water
(b) Adding salt to water
(c) Decreasing the volume of the vessel to one-half
(d) Decreasing the temperature of water
One of the assumptions of kinetic theory of gases states that "there is no force of attraction between the molecules of a gas."How far is this statement correct? Is it possible to liquefy an ideal gas? Explain.
The critical temperature (Tc) and critical pressure (Pc) of C02 are 30.98 °C and 73 atm respectively. Can C02(g) be liquefied at 32 °C and 80 atm pressure?
Critical temperature for Co2 and CH4 are 31.1 °C and -81.9 °C respectively. Which of these has stronger intermolecular forces and why ?
Explain the effect of increasing the temperature of a liquid on intermolecular forces operating between its particles. What will happen to the viscosity of a liquid if its temperature is increased?
Match the graph between the following variables with their names.
| Column I (Graphs) | Column II (Names) |
| (i) Pressure vs temperature graph at constant molar volume. | (a) Isotherms |
| (ii) Pressure vs volume graph at constant temperature. | (b) Constant temperature curve |
| (iii) Volume vs temperature graph at constant pressure. | (c) Isochores |
| (d) Isobars |
A weather balloon has a volume of 175 dm3 when filled with hydrogen gas at a pressure of 1.0 bar. Calculate the volume of the balloon when it rises to a height where the atmospheric pressure is 0.8 bar. Assume that temperature is constant.
Give an expression for the van der Wools equation. Give the significance of the constants used in the equation. What are their units?
With regard to the gaseous state of matter which of the following statements are correct?
(a) Complete order of molecules (b) Complete disorder of molecules
(c) Random motion of molecules (d) Fixed position of molecules
Under which of the following two conditions applied together, a gas deviates most from the ideal behaviour?
(a) Low pressure (b) High pressure
(c) Low temperature (d) High temperature
How much time would it take to distribute one Avogadro number of wheat grains if 1010 grains are distributed each second ?
State and explain Dalton’s law of partial pressures. Can we apply Dalton's law of partial pressures to a mixture of carbon monoxide and oxygen?
Two different gases ˜A' and ˜9' are filled in separate containers of equal capacity under the same conditions of temperature and pressure. On increasing the pressure slightly, the gas ˜A' liquefies but gas ˜B' does not liquefy even on applying high pressure until it is cooled. Explain this phenomenon.
One of the assumptions of kinetic theory of gases is that there is no force of attraction between the molecules of a gas.
State and explain the evidence that shows that the assumption is not applicable for real gases.
Calculate the volume occupied by 8.8 g of CO2 at 31.1 °C and 1 bar pressure. R = 0.083 bar LK-1 mol-1
(a) What do you mean by’Surface Tension'of a liquid?
(b) Explain the factors which can affect the surface tension of a liquid.
Viscosity of a liquid arises due to strong intermolecular forces existing between the molecules. Stronger the intermolecular forces, greater is the viscosity. Name the intermolecular forces existing in the following liquids and arrange them in the increasing order of their viscosities. Also give reason for the assigned order in one line.Water, Hexane (CH3CH2CH2CH2CH2CH3), Glycerine (CH2OHCH(OH)CH2OH)
Pressure versus volume graphs for a real gas and an ideal gas are shown in the figure.

Answer the following questions on the basis of this graph.
(i) Interpret the behaviour of real gas with respect to ideal gas at low pressure.
(ii) Interpret the behaviour of real gas with respect to ideal gas at high pressure.
(iii) Mark the pressure and volume by drawing a line at the point where real gas behaves as an ideal gas.
Assertion (A): Three states of matter are the result of balance between intermolecular forces and thermal energy of the molecules. .
Reason (R): Intermolecular forces tend to keep the molecules together but thermal energy of molecules tends to keep them apart.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
Assertion (A): At constant temperature, PV vs V plot for real gases is not a straight line.
Reason (R): At high pressure all gases have Z> 1 but at intermediate pressure most gases have Z < 1.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
Assertion (A): The temperature at which vapour pressure of a liquid is equal to the external pressure is called boiling temperature.
Reason (R): At high altitude atmospheric pressure is high.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
Why does sharp glass edge become smooth on heating it up to its melting point in a flame? Explain which property of liquids is responsible for this phenomenon.
2.9 g of a gas at 95 °C occupied the same volume as 0.184 g of hydrogen at 17 °C at the same pressure. What is the molar mass of the gas ?
Use the information and data given below to answer the questions (a) to (c):
• Stronger intermolecular forces result in higher boiling point.
• Strength of London forces increases with the number of electrons in the molecule.
• Boiling point of HF, HC1, HBr and HI are 293 K, 189 K, 206 K and 238 K respectively.
(a) Which type of intermolecular forces are present in the molecules HF, HC1, HBr and HI?
(b) Looking at the trend of boiling points of HC1, HBr and HI, explain out of dipole-dipole interaction and London interaction, which one is predominant here?
(c) Why is boiling point of hydrogen fluoride highest while that of hydrogen chloride lowest?
Pressure of l g of an ideal gas A at 27 °C is found to be 2 bar. When 2 g of another ideal gas B is introduced in the same flask at same temperature, the pressure becomes 3 bar. Find the relationship between their molecular masses.
What are ideal and real gases? Out of CO2 and NH3 gases, which is expected to show more deviation from the ideal gas behaviour?
The magnitude of surface tension of liquid depends on the attractive forces between the molecules. Arrange the following in increasing order of surface tension:
water, alcohol (C2H5OH) and hexane [CH3(CH2)4CH3)].
Isotherms of carbon dioxide at various temperatures are represented in the following figure. Answer the following questions based on this figure.

(i) In which state will C02 exist between the points a and b at temperature T1
(ii) At what point will Co2 start liquefying when temperature is T1?
(iii) At what point will C02 be completely liquefied when temperature is T2?
(iv) Will condensation take place when the temperature is T3
(v) What portion of the isotherm at T1 represent liquid and gaseous C02 at equilibrium?
A mixture of dihydrogen and dioxygen at one bar pressure contains 20% by weight of dihydrogen. Calculate the partial pressure of dihydrogen.
A flask was heated from 27 °C to 227 °C at constant pressure. Calculate the volume of the flask if 0.1 dm3 of air measured at 227 °C was expelled from the flask.
The pressure of a 1:4 mixture of dihydrogen and dioxygen enclosed in a vessel is one atmosphere. What would be the partial pressure of dioxygen?
(a) 0.8 x 105atm
(b) 0.008 Nm-2
(c) 8 x 104 Nm -2
(d) 0.25 atm
Assertion (A): At critical temperature liquid passes into gaseous state imperceptibly and continuously.
Reason (R): The density of liqtiid and gaseous phase is equal to critical temperature.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
Why does the boundary between liquid phase and gaseous phase disappear on heating a liquid up to critical temperature in a closed vessel? In this situation what will be the state of the substance?