

The enthalpy of formation of methane at constant pressure and 300 K is – 78.84 kJ. What will be the enthalpy of formation at constant volume?
The equation representing the enthalpy of formation of methane is:
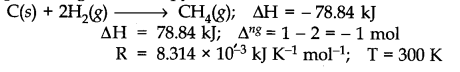

Consider the following reaction between zinc and oxygen and choose the correct options out of the options given below:
2Zn(s) + 02(g) → 2ZnO(s); ∆H=-693.8 kJ mol-1
(i) The enthalpy of two moles ZnO is less than the total enthalpy of two moles of Zn and one mole of oxygen by 693.8 kJ.
(ii) The enthalpy of two moles of ZnO is more than the total enthalpy of two moles of Zn and one mole of oxygen by 693.8 kJ.
(iii) 8 kJ mol -1 energy is evolved in the reaction.
(iv) 693.8 kJ mol-1 energy is absorbed in the reaction.
Give reason for the following:
(a)Neither q nor w is a state function but q + w is a state function.
(b)A real crystal has more entropy than an ideal crystal.
The entropy change can be calculated by using the expression ∆S = q rev / T. When water freezes in a glass beaker, choose the correct statement amongst the following:
When water freezes in a glass beaker, choose the correct statement amongst the following:
(a) ∆S(system) decreases but ∆S(surroundings) remains the same.
(b) ∆S(system) increases but ∆S(surroundings) decreases.
(C) ∆S(system) decreases but ∆S(surroundmgs) increases.
(d) ∆S(system) decreases but ∆S(surroundings) also decreases.
The spontaneity means, having the potential to proceed without the assistance of external agency. The processes which occur spontaneously are
(a) flow of heat from colder to warmer body.
(b) gas in a container contracting into one comer.
(c) gas expanding to fill the available volume.
(d) burning carbon in oxygen to give carbon dioxide.
In an exothermic reaction, heat is evolved, and system loses heat to the surroundings. For such system
(a) qP will be negative
(b) ∆γHwill be negative
(c) qp will be positive
(d) ∆γHwill be positive.
Define the following:
(i) First law of thermodynamics.
(ii) Standard enthalpy of formation.
When two moles of C2H6(g) are burnt, 3129 kj of heat is liberated. Calculate the heat of formation of C2H6(g). ∆fH for C02(g) and H20(l) are-393.5 and -286 kj mol-1 respectively.
Heat capacity (CP) is an extensive property but specific heat (c) is an intensive property. What will be the relation between Cp and c for 1 mol of water?
What is the enthalpy of formation of the most stable form of an element in its standard state?
Calculate the number of kj of heat necessary to raise the temperature of 60 g of aluminium from 35 °C to 55 °C. Molar heat capacity of Al is 24 J mol-1 K-1.
Enthalpy is an extensive property. In general, if enthalpy of an overall reaction A→B along one route is ∆rH and ∆rH1, ∆rH2, ∆rH3 …. represent enthalpies of intermediate reactions leading to product B. What will be the relation between ∆rH for overall reaction and ∆rH1, ∆rH2….. etc. for intermediate reactions.
The enthalpy of atomisation for the reaction CH4(g) → C(g) + 4H(g) is 1665 kJ mol-1. What is the bond energy of C – H bond?
One mole of acetone requires less heat to vapourise than 1 mol of water. Which of the two liquids has higher enthalpy of vapourisation?
1 g of graphite is burnt in a bomb calorimeter in excess of oxygen at 298 K and 1 atmospheric pressure according to the equation C(graphite) + 02 (g) —> C02 (g) During the reaction, temperature rises from 298 K to 299 K. If the heat capacity of the bomb calorimeter is 20.7 kJ/K, what is the enthalpy change for the above reaction at 298 K and 1 atm?
For an ideal gas. the work of reversible expansion under isothermal condition 1.0 mol of an ideal gas is expanded isothermally and reversibly to ten times of its original volume, in two separate experiments. The expansion is carried out at 300 K and at 600 K respectively. Choose the correct option.
can be calculated by using expression w = -nRT In Vf / Vi A sample containing
(a) Work done at 600 K is 20 times the work done at 300 K.
(b) Work done at 300 K is twice the work done at 600 K
(c) Work done at 600 K is twice the work done at 300 K.
(d) ∆U= 0 in both cases.
Expansion of a gas in vacuum is called free expansion. Calculate the work done and the change in internal energy when 1 litre of ideal gas expands isothermally into vacuum until its total volume is 5 litre.
A sample of 1.0 mol of a monoatomic ideal gas is taken through a cyclic process of expansion and compression as shown in the figure. What will be the value of ΔHfor the cycle as a whole?

(a)What is a spontaneous process? Mention the conditions for a reaction to be spontaneous at constant temperature and pressure.
(b) Discuss the effect of temperature on the spontaneity of an exothermic reaction.
In an adiabatic process, no transfer-of heat takes place between system and surroundings. Choose the correct option for free expansion of an ideal gas under adiabatic condition from the following.

Identify the state functions and path functions out of the following: enthalpy, entropy, heat, temperature, work, free energy.
Calculate the enthalpy change on freezing of 1.0 mol of water at 10.0 °C to ice at – 10.0 °C. A, H = 6.03 KJ mot1 at 0 °C. Cp [H20(l)J = 75.3 J mol-1 K-1; Cp [H20(s)J = 36.8 J mol-1 K-1.
Given : N2(g) + 3H2(g) ————> 2NH3(g); ∆r H– = -92.4 kj mot-1 What is the standard enthalpy of formation of NH3 gas?
How are internal energy change, free energy change and entropy change are related to one another?
The bond enthalpy of H2(g) is 436 kj mol-1and that of N2 (g) is 941.3 kj mol-1. Calculate the average bond enthalpy of an N-H bond in ammonia. Given: ∆H– (NH3) = -46 kj mol-1
During complete combustion of one mole of butane, 2658 kJ of heat is released. The thermochemical reaction for above change is

Thermodynamics mainly deals with
(a) interrelation of various forms of energy and their transformation front one from to another.
(b) energy changes in the processes which depend only on initial and final states of the microscopic system containing a few molecules.
(c) how and at what rate these energy transformations are carried out.
(d) the system in equilibrium state or moving from one equilibrium state to another equilibrium state.
Increase in enthalpy of the surroundings is equal to decrease in enthalpy of the system. Will the temperature of system and surroundings be the same when they are in thermal equilibrium?
Enthalpy of combustion of carbon to carbon dioxide is – 393.5 J mol-1 .Calculate the heat released upon formation of 35.2 g of C02 from carbon and oxygen gas.
Enthalpy of sublimation of a substance is equal to
(a) enthalpy of fusion + enthalpy of vapourisation
(b) enthalpy of fusion
(c) enthalpy of vapourisation
(d) twice the enthalpy of vapourisation.