

Predict the products of electrolysis in each of the folloxving:
(i) An aqueous solution of AgNO3 with silver electrodes.
(ii) An aqueous solution of silver nitrate with platinum electrodes.
(iii) A dilute solution of H2S04with platinum electrodes.
(iv) An aqueous solution of CuCl2 with platinum electrodes.
(i) In aqueous solution, AgNO3 ionises to give Ag+(aq) and NO3– (aq) ions.
AgN03(aq) ——–> Ag+(aq) + NO3– (aq)
Thus, when electricity is passed, Ag+(aq) ions move towards the cathode while NO3– ions move towards the anode. In other words, at the cathode, either Ag+(aq) ions or H2O molecules may be reduced. Which of these will actually get discharged would depend upon their electrode potentials which are given below:
Ag+(aq) +e–———-> Ag(s); E ° = +0.80 V …(i)
2H2O(Z) + 2e– ————> H2(g) + 2OH–(aq); E ° = -0.83 V …(ii)
Since the electrode potential (i.e., reduction potential of Ag+(aq) ions is higher than that of H2O molecules, therefore, at the cathode, it is the Ag+(aq) ions (rather than H2O molecules) which are reduced.
Similarly, at the anode, either Ag metal of the anode or H2O molecules may be oxidised. Their electrode potentials are:
Ag(s) ———–> Ag+(aq) + e–; E ° = -0.80 V …(iii)
2H2O(l) ————–> 02(g) +4H+(aq)+4e– ; E ° = -1.23 V …(iv)
Since the oxidation potential of Ag is much higher than that of H2O, therefore,
at the anode, it is the Ag of the silver anode which gets oxidised and not the H2O molecules. It may, however, be mentioned here that the oxidation potential of N03–ions is even lower than that of H2O since more bonds are to broken during reduction of N03 ions than those in H2O.
Thus, when an aqueous solution 0f AgN03 is electrolysed, Ag from Ag anode dissolves while Ag+(aq) ions present in the solution get reduced and get deposited on the cathode.
(ii) If, however, electrolysis of AgN03 solution is carried out using platinum electrodes, instead of silver electrodes, oxidation of water occurs at the anode since Pt being a noble metal does not undergo oxidation easily. As a result, O2 is liberated at the anode according to equation (iv).
Thus, when an aqueous solution of AgNO3 is electrolysed using platinum electrodes, Ag+ ions from the solution get deposited on the cathode while 02 is liberated at the anode.
(iii) In aqueous solution, H2S04ionises to give H+(aq) and SO42-(aq) ions.
H2S04(aq) ——> 2H+(aq) +S04–(aq)
Thus, when electricity is passed, H+ (aq) ions move towards cathode while SO42-(aq) ions move towards anode. In other wode either H+(aq) ions or H2O molecules are reduced. Their electrode potentials are:2H+(aq)2e– ——-> H2(g); E ° = 0.0 V
H2O(aq) + 2e– ——-> H2(g) + 2OH–((aq); E ° = -0.83 V
Since the electron potential (i.e., reduction potential) of H+(aq) ions is higher than that of H2O, therefore, at the cathode, it is H+(aq) ions (rather than H2O molecules) which are reduced to evolve H2 gas.
Similarly at the anode, either SO42-(aq) ions or H2O molecules are oxidised. Since the oxidation potential of SO4 is expected to be much lower (since it involved cleavage of many bonds as compared to those in H20) than that of HjO molecules, therefore, at the anode, it is H2O molecules (rather than SO42- ions) which are oxidised to evolve O2 gas.
From the above discussion, it follows that during electrolysis of an aqueous solution of H2S04 only the electrolysis of H2O occurs liberating H2 at the cathode and O2 at the anode.
(iv) In aqueous solution, CuCl2 ionises as follows:
CuCl2(aq) ——-> CU2+(aq) + 2Cl–(aq)
On passing electricity, CU2+(aq) ions move towards cathode and CU2+(aq) ions move towards anode.
Thus, at cathode, either CU2+(aq) or H2O molecules are reduced. Their electrode potentials are:
CU2++ 2e– ———> Cu(s); E ° = +0.34 V
H2O(l) + 2e– ——–> H2(g) + 2OH–; E ° = -0.83 V
Since the electrode potential of CU2+(aq) ions is much higher than that of H2O, therefore, at the cathode, it is CU2+(aq) ions which are reduced and not H2Omolecules.
Similarly, at the anode, either Cl–(aq) ions or H2O molecules are oxidised. Their oxidation potentials
2Cl–(aq) ——> Cl2(g) + 2e–; AE ° = -1.36 V
2H2O(l) ——>O2(g) + 4H+(aq) + 4e–; ∆E ° = -1.23 V
Although oxidation potential of H2O molecules is higher than that of Cl– ions, nevertheless, oxidation of Cl–(aq) ions occurs in preference to H2O since due to overvoltage much lower potential than -1.36 V is needed for the oxidation of H2O molecules.
Thus, when an aqueous solution of CuCl2 is electrolysed, Cu metal is liberated at the cathode while Cl2 gas is evolved at the anode.
Identify the correct statements with reference to the given reaction.
P4 + 30H– + 3H20→ PH3 + 3H2 P0–2
(a) Phosphorus is undergoing reduction only.
(b) Phosphorus is undergoing oxidation only.
(c) Phosphorus is undergoing oxidation as well as reduction.
(d) Hydrogen is undergoing neither oxidation nor reduction
While sulphur dioxide and hydrogen peroxide can act as an oxidising as well as reducing agents in their reactions, ozone and nitric acid act only as oxidants. Why?
Calculate the oxidation number of phosphorus in the following species.
(a) HPO32- and (b) P043-
In Ostwald’s process for the manufacture of nitric add, the first step involves the oxidation of ammonia gas by oxygen gas to give nitric oxide gas and steam. What is the maximum wight of nitric oxide that can be obtained starting only with 10.0 g of ammonia and 20.0 g of oxygen?
Arrange the following metals in the order in which they displace each other from the solution of their salts.Al, Cu, Fe, Mg and Zn.
Which of the following statement(s) is/are not true about the following decomposition reaction?
2KCIO3 →2KC1 + 302
(a) Potassium is undergoing oxidation.
(b) Chlorine is undergoing oxidation.
(c) Oxygen is reduced.
(d) None of the species are undergoing oxidation or reduction.
Predict the products of electrolysis in each of the folloxving:
(i) An aqueous solution of AgNO3 with silver electrodes.
(ii) An aqueous solution of silver nitrate with platinum electrodes.
(iii) A dilute solution of H2S04with platinum electrodes.
(iv) An aqueous solution of CuCl2 with platinum electrodes.
Which of the following electrodes will act as anodes, when connected to Standard Hydrogen Electrode?
(a) A13-/A1; E °= -1.66 V
(b) Fe2+ /Fe; E °= -0.44 V
(c) Cu2+/ Cu E °=34 V
(d) F2(g)/2F–(aq) E °= 2.87 V
Justify-giving reactions that among halogens, fluorine is the best oxidant and among hydrohalic compounds, hydroiodic add is the best reductant.
(a) Balance the following equation by oxidation number method or by ion electron (half reaction) method.
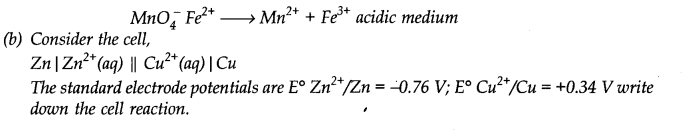
Calculate the oxidation number of each sulphur atom in the following compounds:
(a) Na2S203
(b) Na2S406
(c) Na2S03
(d) Na2S04
Fluorine reacts with ice and results in the change:
H20(S) + F2 (g) ——-> HF(g) + HOF(g)
Justify that this reaction is a redox reaction.
The reaction Cl2(g) + 20H–(aq)→ Cl0–(aq) + Cl–(aq) + H20(l) represents the process of bleaching. Identify and name the species that bleaches the substances due to its oxidizing action.
Depict the galvanic cell in which the reaction, Zn(s) + 2Ag+(aq) ————> Zn2+(aq) + 2Ag(s)
takes place. Further show:
(i) which of the electrode is negatively charged.
(ii) the carriers of current in the cell and
(iii) individual reaction at each electrode.
Which of the following elements does not show disproportionation tendency?
(a) Cl
(b) Br
(c) F
(d) I
Identify the redox reactions out of the following reactions and identify the oxidizing and reducing agents in them

The compound AgF2 is unstable. However, if formed, the compound acts as a very strong oxidising agent. Why?
Identify the substance oxidised, reduced, oxidising agent and reducing agent for each of the following reactions.
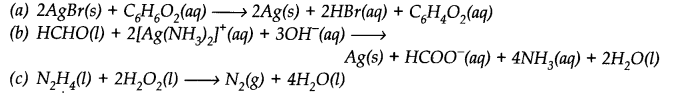
Identify the correct statement(s) in relation to the following reaction:
Zn + 2HCl → ZnCl2 + H2
(a) Zinc is acting as an oxidant.
(b) Chlorine is acting as a reductant.
(c) Hydrogen ion is acting as an oxidant.
(d) Zinc is acting as a reductant.

The exhibition of various oxidation states by an element is also related to the outer orbital electronic configuration of its atom. Atom(s) having which of the following outermost electronic configurations will exhibit more than one oxidation state in its/their compounds.
(a) 3s1
(b) 3dl4s2
(c) 3d24s2
(d) 3s23p3
Write Jour informations about the reaction:
(CN)2(g) + 2OH–(aq) —–> CN–(aq) + CNO–(aq) + H2O(l)
Identify the substance oxidised, reduced, oxidising agent and reducing agent for each of the following reactions.
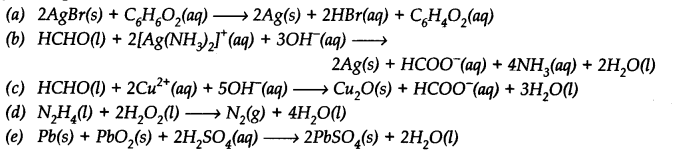
In which of the following compounds, an element exhibits two different oxidation states.
(a) NH2OH
(b) NH4NO3
(c) N2H4
(d) N3H
Write formulas for the following compounds:
(a) Mercury (II) chloride, (b) Nickel (II) sulphate, (c) Tin (IV) oxide, (d) Thallium
(I) sulphate, (e) Iron (III) sulphate, (f) Chromium (III) oxide.
What is standard hydrogen electrode? For what purpose it is used? What are signs of oxidation potential and reduction potential decided by using SHE (Standard hydrogen electrode)?
PbO and Pb02 react with HC1 according to following chemical equations:
2PbO + 4HCl → 2PbCl2 + 2H20
Pb02 + 4HC1 → PbCl2 + Cl2 + 2H20
Why do these compounds differ in their reactivity?
Justify that the following reactions are redox reactions:
(a) CuO(s) + H2(g) —–> Cu(s) + H20(g)
(b) Fe2O3(s) +3CO(g) —-> 2Fe(s) + 3CO2(g)
(c) 4BCl3(g) +3LiAlH4(s) ——> 2B2H6(g) + 3LiCl(s) + 3AlCl3(s)
(d) 2K(s) +F2(g)——> 2K+F–(s)
Consider the reactions:
(a) 6CO2(g) 6H2O(l) ———> C6H12O6(s) + 6O6(g) (b) O3(g) + H2O2(l) H2O(l) + 2O2(g)
Why it is more appropriate to write these reactions as:
(a) 6CO2(g) + 12H2O(l) ————-> C6H12O6(s) + 6H2O(l) + 6O2(g)
(b) O3(g) + H2O2 (l) ———–> H2O(l) + O2(g) + O2(g)
Also suggest a technique to investigate the path of above (a) and (b) redox reactions.
Consider the reactions:

Why does the same reductant, thiosulphate react difforerently with iodine and bromine?
Balance the following redox reactions by ion-electron method.
(a) MnO4–(aq) +I–(aq) ———>Mn02(s) + I2 (s) (in basic medium)
(b) MnO4–(aq) + S02(g) ——-> Mn2+(aq) +H2S04–(in acidic solution)
(c) H2O2(aq) + Fe2+(aq) ———-> Fe3+(aq) + H2O(l) (in acidic solution)
(d) Cr2O72- (aq) + S02 (g)——> Cr3+ (aq) + SO42-(aq) (in acidic solution)
Given the standard electrode potentials,
K+/K = -2.93 V, Ag+/Ag = 0.80 V, Hg2+/Hg = 0.79 V, Mg2+/Mg = -2.37 V,
Cr3+/Cr = -0.74 V. Arrange these metals in increasing order of their reducing power.
What is meant by electrochemical series? What are characteristics of electrochemical series?