

Why does type of overlap given in the following figure not result in the bond formation?

In first figure, the ++ overlap is equal to +- overlap and therefore, these cancel out and net overlap is zero.
In second figure, no overlap is possible because the two orbitals are perpendicular to each other.
Match the species in Column I with the geometry/shape in Column II.
| Column I | Column II |
| (i) H30+ | (a) Linear |
| (ii) HC = CH | (b) Angular |
| (iii) Cl0–2 | (c) Tetrahedral |
| (iv) NH+4 | (d) Trigonal bipyramidal |
| – | (e) Pyramidal |
Which of the following statements are correct about CO32- ?
(a) The hybridization of central atom is sp3.
(b) Its resonance structure has one C – O single bond and two C = O double bonds.
(c) The average formal charge on each oxygen atom is 0.67 units.
(d) All C – O bond lengths are equal.
Account for the following:
(i) Water is a liquid while H2S is a gas
(ii) NH3 has higher boiling point than PH3.
Using molecular orbital theory, compare the bond energy and magnetic character of 0+2 and O–2
Match the shape of molecules in Column I with the type of hybridization in Column II.
| Column I | Column II |
| (i) Tetrahedral | (a) sp2 |
| (ii) Trigonal | (b) sp |
| (iii) Linear | (c) sp3 |
Which hybrid orbitals are used by carbon atoms in the following molecules?
(a) CH3-CH3 (b) CH3-CH = CH2 (c) CH3-CH2-OH (d) CH3-CHO (e) CH3COOH.
In which of the following substances will hydrogen bond be strongest?
(a) HCl
(b) H20
(c) HI
(d) H2S
The skeletal structure of CH3COOH as shown below is correct, but some of the bonds are shown incorrectly. Write the correct Lewis structure for acetic acid.
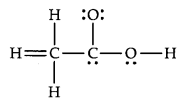
Name the two conditions which must be satisfied for hydrogen bonding to take place in a molecule.
3PO3 can be represented by structures 1 and 2 shown below. Can these two structures be taken as the canonical forms of the resonance hybrid representing H3PO3? If not, give reasons for the same.
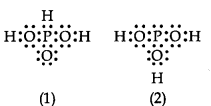
Draw diagrams showing the formation of a double bond and a triple bond between carbon atoms in C2 H4 and C2 H2 molecules.
What is meant by hybridisation of atomic orbitals? Describe the shapes of sp, sp2, sp3 hybrid orbitals.
Describe the change in hybridisation (if any) of the Al atom in the following reaction. AlCl3 + Cl– ——>AlCl4- .
Define Lattice energy. How is Lattice energy influenced by (i) Charge on the ions (ii) Size of the ions?
(a) How many a and n bonds are present in
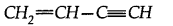
(b) Why Hf is more stable than H2?
(c) Why is B2 molecule paramagnetic?
In N0–3 ion, the number of bond pairs and lone pairs of electrons on nitrogen atom are
(a) 2, 2 (b) 3, 1 (c) 1,3 (d) 4, 0
The energy of σ2pz: molecular orbital is greater than 2px and 2pv molecular orbitals in nitrogen molecule. Write the complete sequence of energy levels in the increasing order of energy in the molecule. Compare the relative stability and the magnetic behaviour of the following species:
N2, N+2, N–2, N22+
Predict the shapes of the following molecules on the basis of hybridization. BC13, ch4, co2, nh3
What is meant by the term average bond enthalpy? Why there is difference in bond enthalpy of O – H bond in ethanol (C2H5OH) and water?
Discuss the shape of the following molecules using the VSEPR model:
BeCl2, BCl3 , SiCl4, AsF5, H2S, PH3
Is there any change in the hybridisation ofB and N atoms as a result of the following reaction ? BF3 + NH3 ——-> F3 B.NH3
Explain the diamagnetic behaviour of P2 molecule on the basis of molecular orbital theory.
Apart from tetrahedral geometry, another possible geometry for CH4 is square planar with the four H atoms at the comers of the square and the C atoms at its centre. Explain why CH4 is not square planar?
In which of the following molecule/ion all the bonds are not equal?
(a) XeF4
(b) BF–4
(c) C2H4
(d) SiF4
Explain the non linear shape of H2S and non planar shape of PCl3 using valence shell electron pair repulsion theory.