

Distinguish between :
(i) Hexagonal and monoclinic unit cells
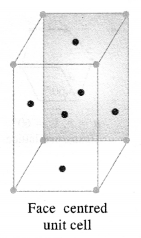
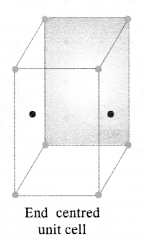
(ii) Face-centred and end-centred unit cells.
i) In a hexagonal unit cell :
a = b # c; α = β = 90° and γ = 120°
In a monoclinic unit cell :
a # b # c and α = γ = 90° and β # 90°
(ii) In a face-centered unit cell, constituent particles are located at all the corners as well as at the centres of all the faces.
In end-centered unit cell, constituent particles are located at all the corners as well as at the centres of two opposite faces. (C.B.S.E Foreign 2015)
Explain how vacancies are introduced in an ionic solid when a cation of higher valence is added as an impurity in it.
Classify each of the following as being either a p-type or n-type semiconductor :
The total number of tetrahedral voids in the face centered unit cell is
(a) 6 (c) 10
(b) 8 (d) 12
In which of the following arrangements octahedral voids are formed?
(a) hep (b) bcc (c) simple cubic (d) fee
If the radius of the octahedral void is r and radius of the atoms in close-packing is R, derive relation between rand R.
Cations are present in the interstitial sites in
(a) Frenkel defect (b) Schottky defect
(c) vacancy defect (d) metal deficiency defect .
Gold (atomic radius = 0.144 nm) crystallises in a face centred unit cell. What is the length of the side of the unit cell ?
Ionic solids, which have anionic vacancies due to metal excess defect, develop colour. Explain with the help of a suitable example.
A sample of ferrous oxide has actual formula Fe0.93 O1.00. In this sample, what fraction of metal ions are Fe2+ ions? What is the type of non-stoichiometric defect present in this sample? '
Ionic solids, which have anionic vacancies due to metal excess defect, develop colour. Explain with the help of a suitable example.
Classify the following as amorphous or crystalline solids: Polyurethane, naphthalene, benzoic acid, Teflon, potassium nitrate, cellophane, polyvinyl chloride, fibreglass, copper
What type of defect can arise when a solid is heated? Which physical property is affected by it and in what way?
Niobium crystallises in a body centred cubic structure. If density is 8.55 g cm-3, calculate atomic radius of niobium, using its atomic mass 93u.
Which of the following is not a characteristic of a crystalline solid?
(a) Definite and characteristic heat of fusion
(b) Isotropic nature
(c) A regular periodically repeated pattern of arrangement of constituent particles in the entire crystal
(d) A true solid
The value of magnetic moment is zero in the case of antiferromagnetic substances because the domains …
(a) get oriented in the direction of the applied magnetic field
(b) get oriented opposite to the direction of the applied magnetic field
(c) are oppositely oriented with respect to each other without the application of magnetic field
(d) cancel out each other's magnetic moment
Assertion (A): Semiconductors are solids with conductivities in the intermediate range from

Reason (R): Intermediate, conductivity in semiconductor is due to partially filled valence band.
Explain how many portions of an atom located at
(i)corner and (ii)body centre of a cubic unit cell is part of its neighbouring unit cell.
Copper crystallises into a fee lattice with edge length 3.61 x 10-8 cm. Show that the calculated density is in agreement with its measured value of 8.92 gcm-3.
Ferric oxide crystallises in a hexagonal dose- packed array of oxide ions with two out of every three octahedral holes occupied by ferric ions. Derive the formula of the ferric oxide.
What type of substances would make better permanent magnets, ferromagnetic or ferrimagnetic. Justify your answer.
Classify each of the following solids as ionic, metallic, modular, network (covalent) or amorphous:
(i) Tetra phosphorus decoxide (P4O10) (ii) Ammonium phosphate, (NH4)3P04 (iii) SiC (iv) I2 (v) P4 (vii) Graphite (viii), Brass (ix) Rb (x) LiBr (xi) Si
How will you distinguish between the following pairs of terms:
(i) Cubic close packing and hexagonal close packing?
(ii) Crystal lattice and unit cell?
(iii) Tetrahedral void and octahedral void?
Which of the following statement is not true about the hexagonal close packing?
(a) The coordination number is 12
(b) It has 74% packing efficiency
(c) Tetrahedral voids of the second layer are covered by the spheres of the third layer
(d) In this arrangement, spheres of the fourth layer are exactly aligned with those of the first layer.
In the cubic close packing, the unit cell has
(a) 4 tetrahedral voids each of which is shared by four adjacent unit cells
(b) 4 tetrahedral voids within the unit cell
(c) 8 tetrahedral voids each of which is shared by four adjacent unit cells
(d) 8 tetrahedral voids within the unit cells.
Under the influence of electric field, which of the following statements are true about the movement of electrons and holes in a p-type semiconductor?
(a) Electron will move towards the positively charged plate through electron holes
(b) Holes will appear to be moving towards the negatively charged plate
(c) Both electrons and holes appear to move towards the positively charged plate
(d) Movement of electrons is not related to the movement of holes
What is the two-dimensional coordination number of a molecule in a square close-packed layer?
Wh ich of the following lattices has the highest packing efficiency (i) simple cubic (ii) body-centered cubic and (iii) hexagonal close-packed lattice?
Niobium crystallises in a body centred cubic structure. If density is 8.55 g cm-3, calculate atomic radius of niobium, using its atomic mass 93u.
Aluminium crystallises in a cubic close packed structure. Its metallic radius is 125 pm.
(i) What is meant by the term coordination number’?
(ii) What is the coordination number of atom
(a) in a cubic close-packed structure?
(b) in a body centred cubic structure?
Iodine molecules are held in the crystals lattice by
(a) London forces (b) dipole-dipole interactions
(c) covalent bonds (d) coulombic forces
In which of the following structure coordination number for cations and anions in the packed structure will be same?
(a) Cl– ions form fee lattice and Na+ ions occupy all octahedral voids of the unit cell.
(b) Ca2+ ions form fee lattice and F- ions occupy all the eight tetrahedral voids of the unit cell
(c) O2- ions form fee lattice and Na+ ions occupy all the eight tetrahedral voids of the unit cell
(d) S2- ions form fee lattice and Zn2+ ions go into alternate tetrahedral voids of the unit cell.
An excess of potassium ions makes KCl crystals appear violet or Lilac in colour since
(a) some of the anionic sites are occupied by an unpaired electron
(b) some of the anionic sites are occupied by a pair of electrons
(c) there are vacancies at some’anionic sites
(d) F-centres are created which impart colour to the crystals
The number of tetrahedral voids per unit cell in NaCl crystal is
(c) twice the number of octahedral voids
(d) four times the number of octahedral voids
Silver crystallises in fcc lattice. If edge length of the cell is 4.07 x 10-8 cm and density is 10.5 g cm-3, calculate the atomic mass of silver.
What is the two-dimensional coordination number of a molecule in square close-packed layer?
Which of the following arrangement shows schematic alignment of magnetic moments of antiferromagnetic substances?

What is the coordination number in a square close packed structure in two dimensions? (a) 2 (b) 3 (c) 4 (d) 6
Amorphous solids can also be called
(a) pseudo solids (b) true solids
(c) super cooled liquids (d) super cooled solids
A perfect crystal of silicon (fig) is doped with some elements as given in the options. Which of these options show n-type semiconductors?


Why does the electrical conductivity of semiconductors increase with rise in temperature?
In the following questions, a statement of Assertion (A) followed by a statement of Reason (R) is given. Choose the correct answer out of the following choices.
(a) Assertion and Reason both are correct statements and Reason is the correct explanation for Assertion.
(b) Assertion and Reason both are correct statements but Reason is not the correct explanation for Assertion.
(c) Assertion is correct but Reason is wrong.
(d) Assertion is wrong but Reason is correct.
Assertion (A): The total number of atoms present in a simple cubic unit cell is one.
Reason (R): Simple cubic unit cell has atoms at its comers, each of which is shared between eight adjacent unit cells.
Assertion (A): The packing efficiency is maximum for the fee structure. Reason (R): The coordination number is 12 in fee structures.
Ionic solids, which have anionic vacancies due to metal excess defect, develop colour. Explain with the help of a suitable example.