

Define antibonding molecular orbital.
The molecular orbital formed by the subtractive effect of the electron waves of the combining atomic orbitals, is called antibonding molecular orbital.
Elements X, Y and Z have 4, 5 and 7 valence electrons respectively, (i) Write the molecular formula of the compounds formed by these elements individually with hydrogen, (ii) Which of these compounds will have the highest dipole moment?
Briefly describe the valence bond theory of covalent bond formation by taking an example of hydrogen. How can you interpret energy changes taking place in the formation of dihydrogen?
What is the effect of the following processes on the bond order in N-, and 02?
(i) N2 → N+2 + e– (ii) 02 → O+2 + e–
Which of the following statements are not correct?
(a) NaCl being an ionic compound is a good conductor of electricity in the solid state.
(b) In canonical structures there is a difference in the arrangement of atoms.
(c) Hybrid orbitals form stronger bonds than pure orbitals.
(d) VSEPR theory can explain the square planar geometry of XeF4.
Match the items given in Column I with examples given in Column II.
| Column I | Column II |
| (i) Hydrogen bond | (a) C |
| (ii) Resonance | (b) LiF |
| (iii) Ionic solid | (c) H2 |
| (iv) Covalent solid | (d) HF |
| (e) 03 |
Structures of molecules of two compounds are given below:

(a) Which of the two compounds will have intermolccular hydrogen bonding and which compound is expected to show intramolecular hydrogen bonding?
(b) The melting point of a compound depends on. among other things, the extent of hydrogen bonding. On this basis explain which of the above two compounds will show higher melting point.
(c) Solubility of compounds in water depends on power to form hydrogen bonds with water. Which of the above compounds will form hydrogen bond with water easily and be more soluble in it?
Draw diagrams showing the formation of a double bond and a triple bond between carbon atoms in C2 H4 and C2 H2 molecules.
Is there any change in the hybridisation ofB and N atoms as a result of the following reaction ? BF3 + NH3 ——-> F3 B.NH3
Diamagnetic species are those which contain no unpaired electrons. Which among the following are diamagnetic?
(a) N2
(b) N22-
(c) 02
(d) o22-
Using molecular orbital theory, compare the bond energy and magnetic character of 0+2 and O–2
Assertion (A): Though the central atom of both NH3 and H20 molecules are sp3 hybridised, yet H – N – H bond angle is greater than that of H – O – H.
Reason (R): This is because nitrogen atom has one lone pair and oxygen atom has two lone pairs.
(a) A and R both are correct, and R is the correct explanation of A.
(b) A and R both are correct, but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A and R both are false.
Arrange the bonds in order of increasing ionic character in the molecules: LiF, K2O, N2, SO2 and ClF3.
Write Lewis structure of the following compounds and show formal charge on each atom. HN03, No2, H2so4
3PO3 can be represented by structures 1 and 2 shown below. Can these two structures be taken as the canonical forms of the resonance hybrid representing H3PO3? If not, give reasons for the same.
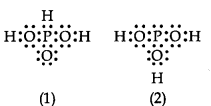
Define Lattice energy. How is Lattice energy influenced by (i) Charge on the ions (ii) Size of the ions?
Which molecule/ion out of the following does not contain unpaired electrons?
(a) N+2
(b) 02
(c) O22-
(d) B2
What is an ionic bond? With two suitable examples explain the difference between an ionic and covalent bond?
Draw the Lewis structures for the following molecules and ions:
H2S, SiCl4 , BeF2, C032-, HCOOH
Although geometries of NH3 and H20 molecules are distorted tetrahedral, bond angle in water is less than that of ammonia. Discuss.
What is the total number of sigma and pi bonds in the following molecules?
(a) C2 H2 (b) C2 H4
Considering X-axis as the intemuclear axis which out of the following will not form a sigma bond and why? (a) Is and Is (b) Is and 2px (c) 2py and 2py (d) Is and 2s
What do you understand by bond pairs and lone pairs of electrons? Illustrate by giving one example of each type.
Write the Lewis dot symbols of the following elements and predict their valencies. (i) Cl (ii) P